| |
The earth's atmosphere has been described by orbiting astronauts as a thin blue line of haze  clinging tenuously to the surface of the planet. From the ground, however, the air appears to us to be rather thick, exerting an average of 14 pounds per square inch on all of its inhabitants, save for the creatures of the seas. It extends up to 50 miles above the surface and each layer clinging tenuously to the surface of the planet. From the ground, however, the air appears to us to be rather thick, exerting an average of 14 pounds per square inch on all of its inhabitants, save for the creatures of the seas. It extends up to 50 miles above the surface and each layer  has characteristics that allow for circulation of gasses and particles between the surface and the upper stratosphere. Its composition is remarkably uniform at the earth's surface, consisting of 20% oxygen, 76% nitrogen and 0.2% carbon dioxide, with traces of argon, methane, and ozone. This last component, O3, is created by the interaction of ultraviolet radiation from the sun with O2 in the upper layers of the stratosphere. This is an essential component of the atmosphere that accumulates in large amounts at each pole during their 6 months of sunlight, and then becomes distributed throughout the upper stratosphere by the action of earth's rotation. Ozone acts as a shield against the mutagenic effects of ultraviolet radiation in the 280-320 nm spectrum (i.e., UV-B). This shield has allowed life to crawl up onto the land masses and reproduce. Mutations occur among all life forms at a rate of about 1 in 106 organisms. Direct exposure to the full range of UV-B radiation causes that rate to increase dramatically. We will return to this concept when we discuss the relationship between ultraviolet radiation and skin cancer. The atmosphere's reassuring deep blue tint is due to the absorption spectrum of oxygen that was and still is produced by countless species of plants. An atmosphere of 20% oxygen was achieved some 408-360 million years ago and has remained at that level ever since. Thus, the composition of the earth's atmosphere is biogenically controlled. That life on earth regulates the components of its own atmosphere is the intellectual center piece of the Gaia Hypothesis (Lovelock, 1971). The Gaia Hypothesis further states that, in addition to the composition of the atmosphere, the rest of our planet is what it is today also as the direct result of life on earth remodeling inanimate geomorphic residues (i.e. rocks, etc.) into states that maintain and support their tolerance limits. The earth is thus viewed as a living rheostat that is constantly being regulated by a myriad of microbial species in the earth's crust, marine protists, and a plethora of terrestrial plant and animal life. If the Gaia hypothesis is even partially correct, then maintaining maximum biodiversity for all functional ecosystems becomes a major goal of public health. has characteristics that allow for circulation of gasses and particles between the surface and the upper stratosphere. Its composition is remarkably uniform at the earth's surface, consisting of 20% oxygen, 76% nitrogen and 0.2% carbon dioxide, with traces of argon, methane, and ozone. This last component, O3, is created by the interaction of ultraviolet radiation from the sun with O2 in the upper layers of the stratosphere. This is an essential component of the atmosphere that accumulates in large amounts at each pole during their 6 months of sunlight, and then becomes distributed throughout the upper stratosphere by the action of earth's rotation. Ozone acts as a shield against the mutagenic effects of ultraviolet radiation in the 280-320 nm spectrum (i.e., UV-B). This shield has allowed life to crawl up onto the land masses and reproduce. Mutations occur among all life forms at a rate of about 1 in 106 organisms. Direct exposure to the full range of UV-B radiation causes that rate to increase dramatically. We will return to this concept when we discuss the relationship between ultraviolet radiation and skin cancer. The atmosphere's reassuring deep blue tint is due to the absorption spectrum of oxygen that was and still is produced by countless species of plants. An atmosphere of 20% oxygen was achieved some 408-360 million years ago and has remained at that level ever since. Thus, the composition of the earth's atmosphere is biogenically controlled. That life on earth regulates the components of its own atmosphere is the intellectual center piece of the Gaia Hypothesis (Lovelock, 1971). The Gaia Hypothesis further states that, in addition to the composition of the atmosphere, the rest of our planet is what it is today also as the direct result of life on earth remodeling inanimate geomorphic residues (i.e. rocks, etc.) into states that maintain and support their tolerance limits. The earth is thus viewed as a living rheostat that is constantly being regulated by a myriad of microbial species in the earth's crust, marine protists, and a plethora of terrestrial plant and animal life. If the Gaia hypothesis is even partially correct, then maintaining maximum biodiversity for all functional ecosystems becomes a major goal of public health.
Cloud formation  is essential for controlling and keeping the planet's ambient temperature at around 20°C, since clouds reflect a significant portion (approximately 30%) of solar radiation back into outer space. The total amount of reflected sunlight is referred to as the albedo. Snow and the ice capped polar regions also contribute to the earth's albedo. The absence of clouds would eventually result in wildly fluctuating ambient temperatures, creating an environment that might select a radically different assemblage of creatures to the ones that currently exist. Too many clouds would also alter our current situation, cooling the earth perhaps below the tolerance limits for most terrestrial life forms, and dramatically reducing the rate of photosynthesis worldwide. Thus, understanding the mechanism of cloud formation is key to understanding the patterns of global warming and cooling. How do clouds form? The process starts in the tropical and neotropical oceans where the amount of solar radiation reaching earth is maximum. The sun shines on the ocean's surface day in and day out, evaporating Hugh amounts of water vapor into the lower atmosphere. Throughout the world's oceans numerous species of Coccolithic photosynthetic algae abound, generating dimethyl sulfide as part of their metabolism. is essential for controlling and keeping the planet's ambient temperature at around 20°C, since clouds reflect a significant portion (approximately 30%) of solar radiation back into outer space. The total amount of reflected sunlight is referred to as the albedo. Snow and the ice capped polar regions also contribute to the earth's albedo. The absence of clouds would eventually result in wildly fluctuating ambient temperatures, creating an environment that might select a radically different assemblage of creatures to the ones that currently exist. Too many clouds would also alter our current situation, cooling the earth perhaps below the tolerance limits for most terrestrial life forms, and dramatically reducing the rate of photosynthesis worldwide. Thus, understanding the mechanism of cloud formation is key to understanding the patterns of global warming and cooling. How do clouds form? The process starts in the tropical and neotropical oceans where the amount of solar radiation reaching earth is maximum. The sun shines on the ocean's surface day in and day out, evaporating Hugh amounts of water vapor into the lower atmosphere. Throughout the world's oceans numerous species of Coccolithic photosynthetic algae abound, generating dimethyl sulfide as part of their metabolism.

Clouds as observed from the Space Shuttle 300 mi. above the Earth |
|

Coccolithic algae |
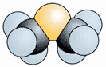
Dimethyl Sulfide,
a volatile metabolic by-product |
| |
|
This algal waste product also vaporizes into the lower atmosphere, mixing with dust particles and water vapor to form micelles. The energetic spectrum of sunlight converts dimethyl sulfide to sulfuric acid which is extremely hygroscopic, trapping larger amounts of water vapor onto the acidified particle's surface. Micelles of dust, sulfuric acid and water, when moved to higher regions of the atmosphere by the earth's rotation, condense into thick mats of clouds as the result of a drop in the temperature. When enough cloud is produced over a given area of ocean, the algae in that region receive less sunlight, photosynthesize less, and the amount of dimethyl sulfide thus goes down. As a result, cloud formation decreases. As clouds drift away from a given area, the sun shines once again, and induces photosynthesis to resume at maximum rates. As a result, clouds begin to form and the process repeats over and over again. This slow, regular oscillation produces a constantly changing cloud cover that, in turn regulates the amount of sunlight striking the earth's surface. The albedo of the earth's surface determines the atmospheric temperature. Before humans evolved, plant life and global patterns of ocean circulation  combined to create an average Earth temperature of 20°C. In addition, ocean currents were largely responsible for the patterns of weather. Recent perturbations in this rheostatic system has resulted in a temperature change that ultimately could have devastating effects on all life on the planet's surface. combined to create an average Earth temperature of 20°C. In addition, ocean currents were largely responsible for the patterns of weather. Recent perturbations in this rheostatic system has resulted in a temperature change that ultimately could have devastating effects on all life on the planet's surface.
Despite the fact that the United States and many other countries have enacted legislation (e.g., The Clean Air Act) to protect us all from the ill effects of polluted air, anthropogenic sources of greenhouse gases [Link2 ] (water vapor, methane, CO2 ] (water vapor, methane, CO2  , NO2), CFCs (chlorofluorocarbons from fluid refrigerants) and particulates have already had measurable effects on human health. For example, the composition and behavior of our atmosphere (eg., carbon cycle , NO2), CFCs (chlorofluorocarbons from fluid refrigerants) and particulates have already had measurable effects on human health. For example, the composition and behavior of our atmosphere (eg., carbon cycle  ) has been greatly altered, raising the average temperature of the earth by 1.5°C over the last 50 years, and seasonally depleting the protective ozone layer in the stratosphere above the poles and certain areas of Europe and North America. What are the health consequences of these changes? Do changes in some remote ecosystems resulting from ozone depletion impact our daily lives (e.g., decreases in populations of Southern Ocean phytoplankton)? If so, what can anyone do about these global problems? ) has been greatly altered, raising the average temperature of the earth by 1.5°C over the last 50 years, and seasonally depleting the protective ozone layer in the stratosphere above the poles and certain areas of Europe and North America. What are the health consequences of these changes? Do changes in some remote ecosystems resulting from ozone depletion impact our daily lives (e.g., decreases in populations of Southern Ocean phytoplankton)? If so, what can anyone do about these global problems?
Readings:
Lovelock, J.E. & J.P. Lodge. “Oxygen-the Contemporary Atmosphere”. Atmospheric Environments. Vol. 6. pp. 575-578.
Lovelock, J.E. & Margulis, L. 1975. “The Atmosphere as Circulatory System of the Biosphere: The Gaia Hypothesis”. CoEvolution Quarterly. Summer. pp. 30-41.
Dixon, B. Unseen Power: How Microbes Rule the World. New York City: Oxford University Press, 1995.
Leffell, D.J. & Brash, D.E. 1996. “Sunlight and Skin Cancer”. Scientific American. July. pp. 38-43.
Brackie, R.M., Marks, R., & Green, A. 1996. “The Melanoma Epidemic: Excess Exposure to Ultraviolet Light is Established as the Major Risk Factor” (letter). British Medical Journal. 312: 1362.
[Back To Top] |
|